Abstract
Background: B-cell precursor acute lymphoblastic leukemia (BCP-ALL) is the most frequent pediatric cancer. The most common numerical and structural chromosomal alterations are high hyperdiploidy (HHD, 51-67 chromosomes) and t(12;21)(p13;q22) leading to ETV6::RUNX1 fusion. However, for a full-blown leukemia, secondary alterations are required. Furthermore, it is increasingly recognized that germline variants may enhance ALL susceptibility in ~4% of the patients (Bloom et al. 2019, Klco & Mullighan 2020). In large-scale genomic studies, the mutational landscape of somatic and germline single nucleotide variants and small indels has been well described in BCP-ALL. However, a comprehensive analysis of somatic and germline structural variants (SVs) in ETV6::RUNX1 and HHD tumors is still lacking. Recent feasibility studies showed that whole genome optical mapping (WGOM) is a novel method for reliable SV detection which could abrogate conventional cytogenetics and array based methods in the future (Neveling et al. 2021, Mantere et al. 2021).
The aim of the study was to analyze the structural variation landscape in pediatric BCP-ALL patients by employing WGOM to gain significant insights on leukemia predisposition and development.
Methods: We studied 60 pediatric patients diagnosed with ETV6::RUNX1 (n=30) or HHD (n=30) BCP-ALL with a median age at diagnosis of 3.9 (range 1.7-17) and a male to female ratio of 1.14:1. We collected tumor material at initial diagnosis and included fibroblast or remission material as matched non-tumor control of each patient. Ultra-high molecular weight DNA was extracted from 120 samples and labeled by a sequence specific enzyme. Fluorescently labeled DNA was analyzed using a Saphyr instrument (Bionano Genomics). We integrated comprehensive datasets of 60 matched tumor/non-tumor genomes to identify somatically acquired SVs >500bp. Corresponding data of standard methods as cyto- (karyotype, fluorescence in situ hybridization) and molecular genetics (SNP-array) of the leukemia samples were compared to WGOM data. In addition, the prevalence of rare germline SVs (<1% of 160 healthy control samples [Bionano] and minor allele frequency <0.01 in dbVar) occurring in known cancer predisposition genes was analyzed in the non-tumor samples.
Results: WGOM was able to detect 94% (438/464) of previously observed events by conventional methods, including ETV6::RUNX1 and high hyperdiploid hallmark alterations in all BCP-ALL cases. Beyond that, we discovered 895 additional SVs with WGOM not detected by standard cyto- and molecular genetics.
WGOM detected a median of 25 (range 0-70) somatic SVs secondary to the hallmark alterations in ETV6::RUNX1 tumors compared to 13 (range 0-28) SVs in HHD cases. We identified 60 recurrently altered regions occurring in ≥3 BCP-ALL cases including 12p13.2 (ETV6, 19/60), 9p13.2 (PAX5, 13/60), 9p21.3 (CDKN2A/B, 12/60), 11p12 (RAG2, 10/60), 20p12.2 (MKKS/SLXIP, 10/60) or 3q13.2 (CD200/BTLA, 8/60). We detected ETV6::RUNX1 subtype specific recurrent focal deletions in ATF7IP (10/30), BTG1 (10/30), NSD2 (5/30) and TBL1XR1 (4/30).
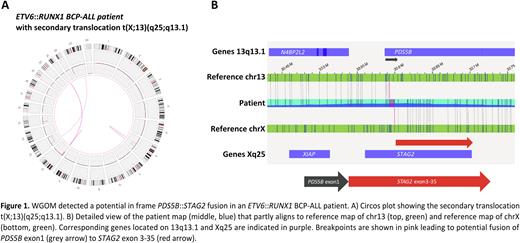
Interestingly, we discovered recurrent SVs affecting putative novel genes involved in BCP-ALL leukemogenesis such as STAG2, GPN3 and UBA2 which are affected in ~10% of the BCP-ALL patients. Strikingly, WGOM revealed in total 9 novel potential in-frame fusion transcripts in both tumor subtypes, including a potential fusion of the cohesin complex members PDS5B::STAG2, arising through a t(X;13)(q25;q13.1) in an ETV6::RUNX1 patient (Figure 1).
In addition, we detected 28 rare germline SVs targeting 22 known cancer predisposing genes in 35% (21/60) of the pediatric patients. 40% (9/22) of the affected genes were involved in hematopoietic or lymphoid organ development as ETV6, FANCA, IKZF1, ITCH or PTPRC. In 18% (11/60) of the children, the identified germline alteration potentially altered a coding region of a known cancer predisposing gene including a duplication of CIITA in an ETV6::RUNX1 patient.
Conclusions: By applying the novel technology WGOM, we could detect a significant amount of previously hidden SVs in our BCP-ALL cohort in addition to conventional methods. We further generated a comprehensive landscape of somatic and germline SVs in childhood BCP-ALL, revealing recurrently altered regions and novel potential players in ALL leukemogenesis and predisposition.
Disclosures
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal